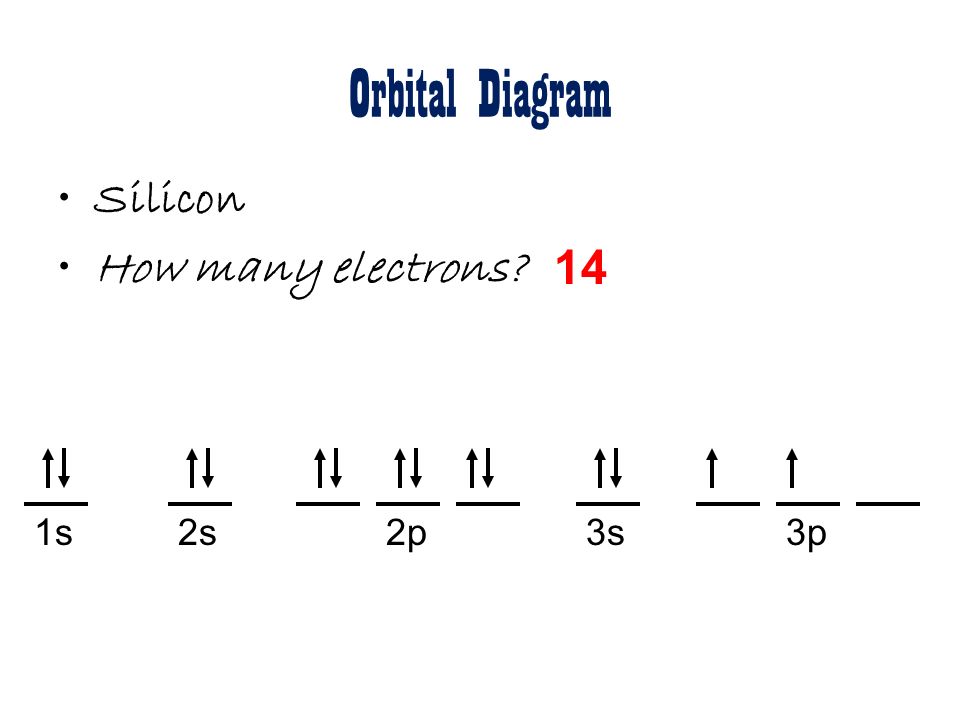
  In practice, chemists simplify the notation by using a bracketed noble gas symbol to represent the configuration of the noble gas from the preceding row because all the orbitals in a noble gas are filled. When we reach neon, with Z = 10, we have filled the 2 p subshell, giving a 1 s 22 s 22 p 6 electron configuration:Īs we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner subshells. Similarly, fluorine has the electron configuration 1 s 22 s 22 p 5: Fluorine has one electron pair in the 1 s orbital, one electron pair in the 2 s orbital, and 2 electrons pairs with one unpaired electron in the 2 p orbital. Electron configuration of Silicon is Ne 3s2 3p2. In the periodic table, the elements are listed in order of increasing atomic number Z. Newton) Starting at hydrogen and the 1s subshell, read across each row of the periodic table until you get to your chosen element. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. Oxygen has one electron pair in the 1 s orbital, one electron pair in the 2 s pair, and one electron pair, and 2 unpaired electrons in the 2 p orbitals. 1: Periodic table of the elements with the location of sodium (Na) highlighted. It has four valance electrons like Carbon. 1s2 2s2 2p6 3s2 3p2 Silicon has a similar electron configuration to Carbon. The outer valance electron configuration of s2 p2 allows Silicon to have common charges of +2 and +4. Si: Melting Point: 1414 ☌: Atomic Weight: 28. The portion of Silicon configuration that is equivalent to the. It has four valance electrons like Carbon. The ground state abbreviated electronic configuration of Neutral Silicon atom is Ne 3s2 3p2. In the third period the 3s subshell is filling for Na and Mg, and therefore Al, Si, P, S, Cl, and Ar. Because all the 2 p orbitals are degenerate, it doesn’t matter which one has the pair of electrons. Explanation: Silicon has a similar electron configuration to Carbon. One electron must be paired with another in one of the 2 p orbitals, which gives us two unpaired electrons and a 1 s 22 s 22 p 4 electron configuration. This negative silicon ion(Si 4-) has fourteen protons, fourteen neutrons, and eighteen electrons. Here, the electron configuration of silicon ion(Si 4-) is 1s 2 2s 2 2p 6 3s 2 3p 6. Silicon is the 14th element of the periodic table so its atomic number is 14. The electron configuration of nitrogen is thus 1 s 22 s 22 p 3.Īt oxygen, with Z = 8 and eight electrons, we have no choice. Silicon is a classified metalloid element and its symbol is Si. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
